probesOur laboratory investigates the methods for molecular imaging, identification of biological molecules, and light control of enzymatic activities in complex systems of biological interest. For this purpose, we are using novel techniques of basic chemistry and protein engineering.
We are studying to discover new principles and develop new methods for spatiotemporal analysis of biological molecules and signal transductions in living organisms. Raman microscopeThe goals of our research activities are 1) to develop analytical methods for visualizing the functions and dynamics of specific biological molecules in living cells and living subjects using fluorescent and bioluminescent proteins, 2) to develop methods for identifying new molecular species involved in intracellular signaling from chemical and cDNA libraries, and 3) to create functional proteins that control the activities of target proteins with external light. We are also striving to develop analytical reagents and methodologies that will lead to the discovery of new biological phenomena through the knowledge of protein structures and functions. Our research will contribute not only to basic research into the mechanisms of molecules in the field of basic biology, but also to new drug development and medical diagnoses.


1) Methods of imaging biomolecular functions and dynamics
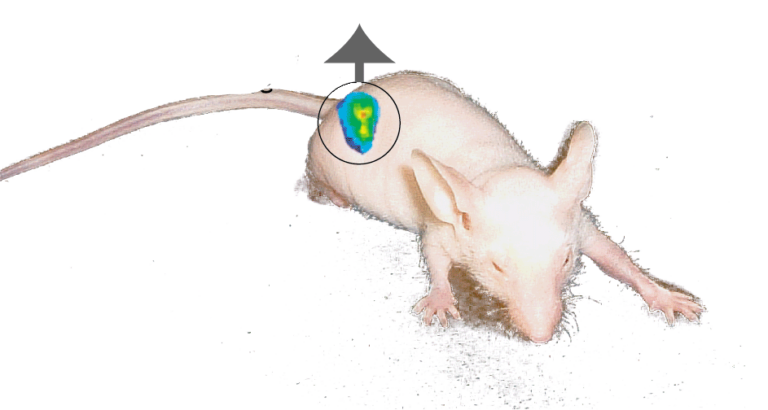
TIRF microscopeRecent advances in analytical methods and technologies, including capillary electrophoresis and mass spectrometry, have made great contributions to the dramatic progress in biological research. It should be noted, however, that conventional analyses are for the most part destructive or invasive because they need cell lysates before detection of a target molecule. The “true physiological functions” of biomolecules could be elucidated if their functions ms imageand dynamics would be visualized noninvasively in living cells and subjects. We are engaged in developing new methods and technical innovations for temporally visualizing the functions and dynamics of biomolecules, so as to determine when, where, and how much intracellular small molecules, RNAs, and proteins exhibit their functions. Methods that we have developed to date include fluorescence imaging of a specific intracellular RNA, an analysis for protein-protein interactions and protein phosphorylation, and a method for imaging the intracellular dynamics of proteins in living animals. A novel microscopic system for imaging biomolecules in living subjects is also under development.


2) Methods for identifying novel molecules involved in an intracellular signaling
The identification of biomolecules such as proteins and lipids depends largely on the development of screening methods. A new screening method would possibly lead to the discovery and identification of novel molecular species. ms imageWe developed our own technology for high-throughput screening of a broad range of libraries for proteins and peptides with desired functions, and also developed a method for comprehensive analyses of proteins transported into mitochondria and endoplasmic reticulum. We also succeeded in determining the signal sequence by screening many peptide libraries, which can lead proteins into mitochondrial intermembrane space (IMS). Currently, we are working to develop a library screening method based on a new principle, and to discover molecular species having unusual functions from the currently available wide variety of chemical and genetic libraries. We are planning to make use of the newly-discovered molecules to elucidate new biological phenomena.
3) Development of optogenetic tools that control biological activities
A calcium chelating reagent, Ethylenediaminetetraacetic acid (EDTA), is important chemicals for understanding the function of calcium ion in living systems because of controlling its ion activities. Likewise, provided that the quantity and functionality of a target protein can be controlled chemically or physically inside the cells, important findings concerning the function will be elucidated in living subjects. ms imageCurrently, functional control of biomolecules is achieved using a small chemical compound such as the inhibitors of enzymes. If light control of the function might become possible, biomolecules that function in living subjects, as well as in the cells, could be regulated both temporally and spatially. We are studying to establish a new technology for controlling biomolecular functions in time and space by means of external light.